Exciting Milestone: Lugano Team Recruits First Patient to MultiSCRIPT
We are excited to share some fantastic progress in our MultiSCRIPT recruitment efforts!
As we reach the sixth month of our recruitment phase, we are pleased to announce that the team in Lugano has successfully recruited their first patient to the MultiSCRIPT study. This marks a significant milestone, and we extend our warmest congratulations and heartfelt thanks to the dedicated Lugano team for their hard work and perseverance.
As of July 5, 2024, we have recruited a total of 286 SMSC participants to the MultiSCRIPT study. This impressive figure represents over 30% of our target recruitment, achieved in just five months. This accomplishment is a testament to the relentless efforts of all our recruiting physicians and the incredible commitment of the SMSC participants. We are deeply grateful to each of you for your contributions and dedication.
We would like to give special recognition to Chiara Zecca, Claudio Gobbi, and Giulio Disanto for their unwaverYour efforts have been instrumental in driving our success so far.
Looking ahead, we will continue to initiate other centers throughout the summer, expanding our reach and furthering our recruitment goals. As we progress, we remain committed to advancing our research and making meaningful strides in our study.
Stay tuned for more updates as we continue our journey with MultiSCRIPT.
Prof. Cristina Granziera:Radio Interview on World Multiple Sclerosis Day
On May 30, 2024, to mark World Multiple Sclerosis Day, Prof. Cristina Granziera was the focus of an insightful interview on radio SFR1. During this comprehensive discussion, Prof. Granziera was invited as a leading expert in the field, and shared valuable information about the complexities of Multiple Sclerosis (MS), shedding light on its multifaceted nature.
Key Highlights from the SFR1 Interview:
Advancements in Diagnosis: Prof. Granziera explained that MS is an autoimmune disease where the immune system attacks the myelin layers in the brain. Over the past two decades, significant progress in imaging and the development of new biomarkers has improved the accuracy of MS diagnosis. This has led to an increase in the number of diagnosed cases, as more subtle signs of the disease can now be detected.
Increased Longevity and Quality of Life: One of the reasons for the rise in MS diagnoses is the improved therapies available today. Patients with MS are living longer and better lives due to these advancements, resulting in a higher number of individuals living with the condition.
Triggers and Causes: Despite intensive research, the exact cause of MS remains unknown. However, Prof. Granziera highlighted that factors such as viral infections, vitamin deficiencies (especially vitamin D), smoking, and genetic predisposition play roles in triggering MS. It is important to note that having a genetic predisposition does not guarantee that an individual will develop MS, as other environmental and lifestyle factors are involved.
Symptoms and When to Seek Medical Advice: MS is often called the "disease of a thousand faces" because its symptoms can vary widely. Common symptoms include visual disturbances, muscle weakness, dizziness, pain, and bladder disorders. Prof. Granziera emphasized the importance of seeking medical advice if any of these symptoms are experienced.
Current and Future Treatments: Currently, there is no cure for MS because the exact cause is still unknown. However, there are around 20 immunomodulating drugs available that can slow the progression of the disease and alleviate clinical symptoms.
Listen to the full interview on radio SFR1 and learn more about the ongoing fight against Multiple Sclerosis.
Profesorship Announcement: Prof. Dr. Lars Hemkens for Clinical Epidemiology
The University Council of the University of Basel has approved the appointment of Prof. Dr. Lars G. Hemkens to a titular professorship at the Faculty of Medicine for Clinical Epidemiology.
Lars G. Hemkens, MD, MPH, is a Senior Scientist at the Department of Clinical Research, University of Basel, and leads Pragmatic Trials and Real World Evidence at the Research Center for Clinical Neuroimmunology and Neuroscience Basel (RC2NB). Additionally, Dr. Hemkens is affiliated with the Meta-Research Innovation Center at Stanford (METRICS; Stanford University, USA).(https://lnkd.in/ecgsCUT7)
Previously, Dr. Hemkens served as the Deputy Director of the Basel Institute for Clinical Epidemiology and Biostatistics (ceb) until its closure. He has also worked at the Stanford Prevention Research Center (Stanford University) and the Institute for Quality and Efficiency in Health Care (IQWiG, Cologne, Germany; Department of the Director). At RC2NB, he leads the workstream Pragmatic Trials and Real World Evidence and the Pragmatic Evidence Lab. (www.pragmatic-evidence.org) His research includes developing and investigating digital biomarkers and digital health applications in multiple sclerosis. Dr. Hemkens is also the Co-PI of MultiSCRIPT, a nationwide pragmatic platform trial fully embedded in the Swiss MS cohort, aimed at continuously improving care for persons with multiple sclerosis.
In his role at the Department of Clinical Research, Dr. Hemkens co-designs various clinical studies across a wide range of medical fields and closely supervises the design of nationwide investigator-initiated clinical trials. He is an associate editor and thematic series editor of “Big data for randomized trials” at the journal Trials, and serves on the editorial board of BMJ Evidence-Based Medicine and the working committee of the RECORD reporting guideline group. Dr. Hemkens also teaches comparative effectiveness research and pragmatic trial design at the University of Basel and Sorbonne Paris Cité, France.
RC2NB is honored to have Dr. Hemkens as a valued member of our team. We support his substantial contributions and eagerly anticipate his ongoing influence in the field of clinical research, neuroscience and digital health.
Link to the University of Basel announcement: here
A novel imaging marker of cortical “cellularity” in multiple sclerosis patients

Numerical simulations show the accuracy of sphere radius estimates in the CONNECTOM scanner (a,c) and on a clinical scanner (b,d). (a,b) “Intra-cellular”: we included spheres, simulating cells, and sticks, simulating neurites, in equal proportion. (c,d) “Intra-cellular and extra-cellular”: here, we added an extra axonal signal simulated with tensor components. In all simulations, Rician noise with SNR = 50 was added. Distance (defined as difference in μm) from the ground truth (GT) is evaluated. Red = overestimation, blue = underestimation, light = acceptable error. Simulation parameters can be found in the “Numerical simulation” in “Methods” section. Back to article page
We are excited to share that our recent research on imaging biomarkers in multiple sclerosis (MS) has been published in Nature Scientific Reports. This collaborative project, led by Muhamed Barakovic and Cristina Granziera, investigates the potential of diffusion MRI and a new model called "soma and neurite density imaging (SANDI)" to non-invasively analyze cellularity in the cortex of individuals with MS.
Paper Key Findings:
- Overall Decrease in Cortical Cellularity: The study revealed a significant reduction in cortical SANDI form in people with MS (pwMS) compared to healthy controls, indicating global degenerative processes consistent with neuronal loss.
- Differences in MS Types: Progressive MS (PMS) showed higher SANDI fsoma in the outer cortical layer, suggesting increased innate inflammatory cells in these regions. Relapsing-remitting MS (RRMS) exhibited increased SANDI fsoma in subpial lesions, aligning with existing pathological data.
- Correlation with sNfL: A significant correlation between SANDI fsoma and serum neurofilament light chain (sNfL), a biomarker of inflammatory axonal damage, indicating a link between SANDI fsoma and inflammatory processes in pwMS.
The data emphasizes the potential of SANDI in clinical settings and its use as a biomarker for monitoring changes in cellularity associated with neurodegeneration and neuroinflammation in MS patients. This research represents a significant advancement in understanding MS pathology and in improving diagnostic and treatment strategies.
Read full article here
Guidance on terminology, application, and reporting of citation searching: the TARCiS statement
We are excited to announce the publication of an important methods guidance paper in the British Medical Journal (BMJ) titled "Guidance on Terminology, Application, and Reporting of Citation Searching: the TARCiS Statement."
Systematic literature searches form the basis of evidence-based healthcare by rigorously identifying relevant studies. While traditional term-based searching in electronic databases is well-established, citation searching has become a crucial technique to discover additional relevant evidence. However, the methodology and terminology associated with citation searching have lacked standardization for decades.
To address this gap, a four-round Delphi consensus study was conducted resulting in the development of the Terminology, Application, and Reporting of Citation Searching (TARCiS) statement. TARCiS offers 10 specific recommendations, each with a detailed rationale and guidance on how and when to conduct and report citation searching in systematic literature reviews. Additionally, the statement identifies four research priorities aimed at further advancing the methodology.
This comprehensive guidance was developed in collaboration with the medical library of the University of Basel, the Eastern Switzerland University of Applied Sciences, and an international panel of 30 experts. The TARCiS statement is expected to significantly enhance the standardization and transparency of citation searching in systematic reviews, encouraging review teams to integrate these recommendations into their workflows.
We encourage researchers, systematic review teams, and healthcare professionals to incorporate the TARCiS recommendations into their practices to improve the quality and consistency of evidence syntheses.
Clinnova - Personalized Medicine with Optimum Data Protection
RC2NB is excited to share the latest media coverage of one of our innovative international projects - Clinnova. This project focuses on revolutionizing autoimmune disease treatment through personalized medicine while ensuring data quality and protection using federated learning - a decentralized approach to AI training.
Clinnova consortium establishes Basel as the center of excellence for the region's multiple sclerosis (MS) treatment and highlights the close collaboration between our team, the University of Basel and the University Hospital Basel. The project, led by our co-CEO, Cristina Granziera, has received CHF 4 million in funding from the Basel Canton, benefiting patients and strengthening Basel's position in the global medical research community.
Project Highlights:
Personalized Treatment: Using AI, Clinnova will analyze large volumes of patient data to tailor therapies to individual needs, enhancing treatment efficacy and reducing side effects.
International Collaboration: The Clinnova consortium ensures data quality and protection through decentralized AI training, keeping patient data within local institutions while sharing insights globally.
Innovative Approach: Federated learning led by Bram Stieltjes will enable the development of algorithms that predict the best treatment options and timing for each patient, especially crucial for diseases like MS, known for its diverse manifestations.
We invite you to join us in sharing the news about this initiative that underscores Basel's role as a hub of medical excellence and innovation. Together, we are paving the way for a future where AI and personalized medicine transform healthcare.
Read the full article written by Agelika Jacobs to learn more about this remarkable project and the future of precision medicine here
Major achievement for the MultiSCRIPT trial

MultiSCRIPT has recently achieved a major milestone with 1st patient enrolled on February 5th, 2024 in Basel. Since then, over 150 participants have been enrolled and randomized in less than 3 months (anticipated recruitment is 915 participants over a 2-year period).
This is a testament that keeping things simple and fully embedded in the patient’s care contributes to a smooth recruitment. MultiSCRIPT is a pragmatic clinical trial that's embedded within the Swiss MS cohort, and it seeks to assess the benefits of 6-monthly sNfL (serum neurofilament light chain) monitoring in addition to usual care compared to usual care alone. The trial aims to provide further evidence and insights into the potential advantages of incorporating sNfL monitoring into routine clinical practices to inform more personalized strategies for multiple sclerosis (MS).
For those who are interested in learning more about MultiSCRIPT-Cycle 1, a detailed protocol is available as a preprinthttps://www.medrxiv.org/content/10.1101/2024.03.22.24304720v1
We encourage you to check it out and share it with others who might find it useful.
We would like to express our gratitude to the study coordinators, nurses, and physicians at the MS center in Basel, whose hard work and dedication have made this achievement possible. Thank you to the Swiss National Science Foundation for supporting this Investigator Initiated Trial.
We are also pleased to announce that we will initiate the other SMSC centers soon, including Ospedale Regionale di Lugano, University Hospital Geneva, University Hospital Lausanne, Inselspital Bern, Kantonsspital Aarau, University Hospital Zürich, and Kantonsspital St. Gallen.
If you're interested in learning more about our awesome team and contributors, we encourage you to visit the multiSCRIPT website at the following URL: https://multiscript.rc2nb.ch
We are excited about the progress and believe that MultiSCRIPT will significantly improve the lives of people with MS.
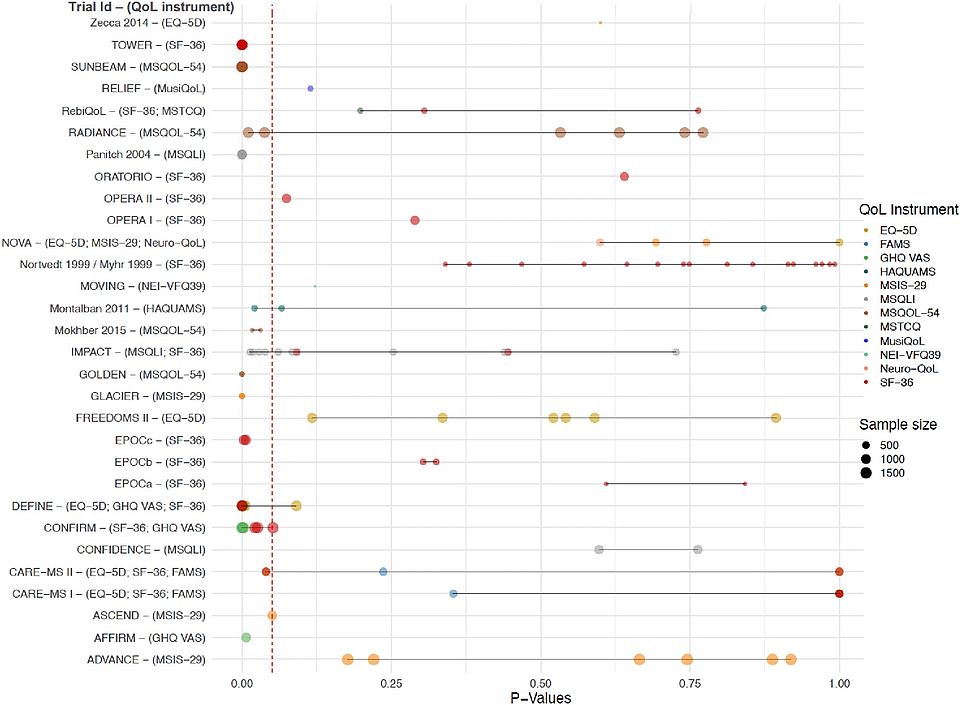
Clinical trial evidence of quality-of-life effects of disease-modifying therapies for multiple sclerosis: a systematic analysis
We are thrilled to unveil our latest review, delving deep into the evidence surrounding the impact of Disease-Modifying Therapies (DMTs) on the quality of life for individuals battling Multiple Sclerosis (MS).
Key findings:
- We analyzed 38 trials spanning from 1999 to 2023, involving over 23,000 participants.
- DMTs have the potential to significantly improve quality of life
- However, there seems to be no generally accepted standard for assessing disease-specific QoL in PwMS, and rarely is this end point in the focus of DMT-evaluating trials.
Join us in spreading awareness about the critical importance of quality of life in MS care. Together, let's advocate for better-designed trials that reflect the needs of patients.
Read full article: Clinical trial evidence of quality-of-life effects of disease-modifying therapies for multiple sclerosis: a systematic analysis
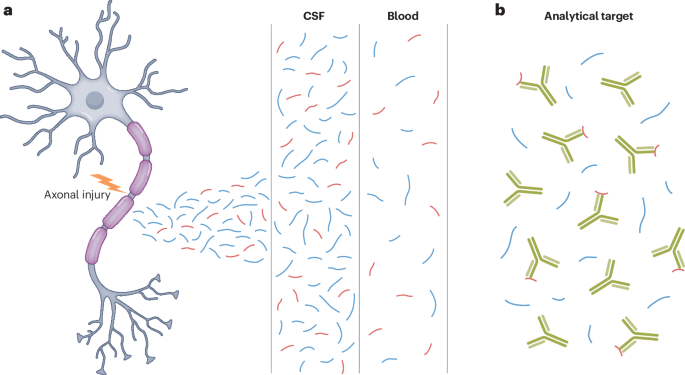
Neurofilaments as biomarkers in neurological disorders — towards clinical application
The latest publication from the RC2NB team published in Nature Reviews Neurology. Neurofilament proteins have emerged as valid biomarkers of neuronal injury and loss. Based on our previous review from 2018 that summarized what was known at the time about neurofilaments as biomarkers for neurological disorders, we here provide an update on the structure and function of neurofilaments, analytical approaches, and challenges in different clinical contexts. This review focuses on the progress made in recent years towards the clinical application of neurofilaments as a biomarker in various neurological disorders.
Definitions of digital biomarkers: a systematic mapping of the biomedical literature
In the newest study published in BMJ Health Care & Informatics, the Pragmatic Evidence team delved into the realm of digital biomarkers, aiming to uncover the essence of this evolving concept. All studies that prominently use the term "digital biomarker" were assessed. However, most of them did not provide a definition, and out of the 128 articles that did provide a definition, 127 used different ones. This indicates a clear lack of consensus in this emerging field. Our overview aims to guide the development of a more harmonized and widely accepted definition.
Read the full publication here: Definitions of Digital Biomarkers
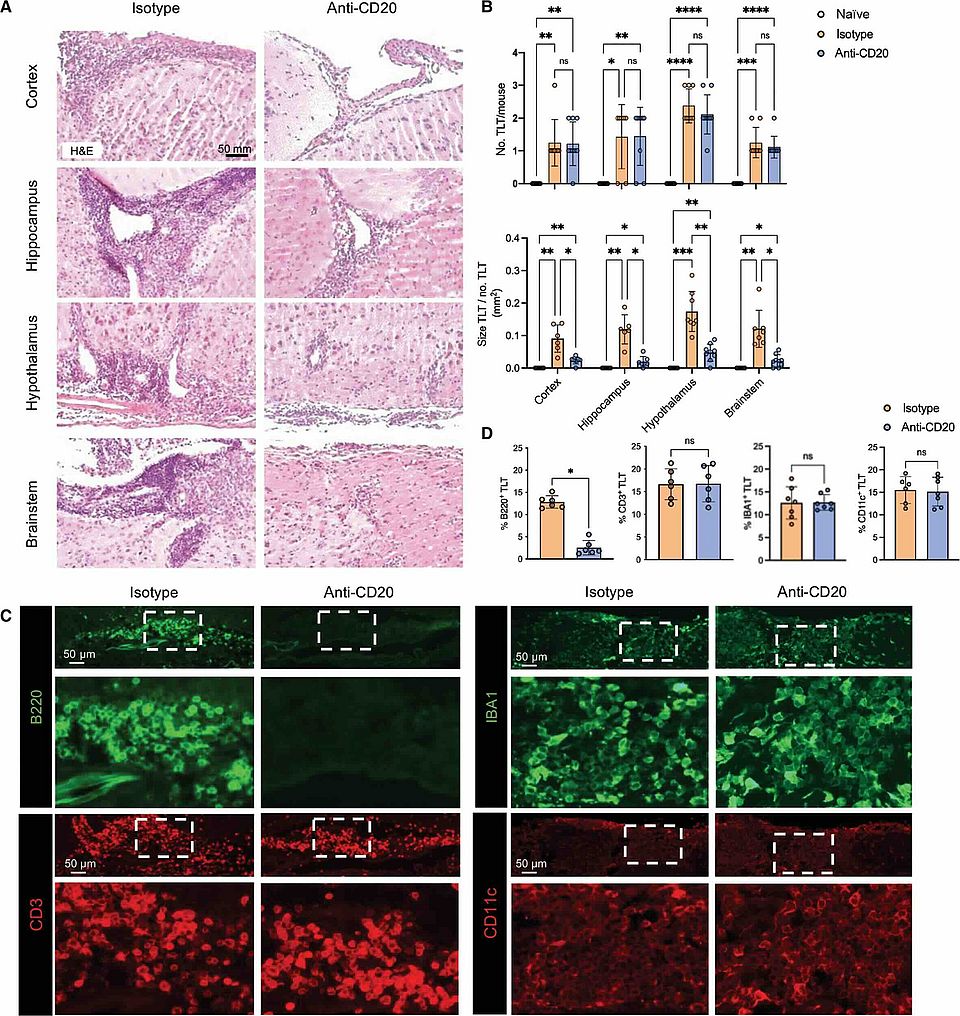
B cell depletion with anti-CD20 promotes neuroprotection in a BAFF-dependent manner in mice and humans
Researchers from the University of Basel and the University of Toronto, Mainz have identified a potential new mechanism by which B cell therapy may protect the brain in MS patients. This study, published in Science Translational Medicine, suggests B cell depletion may safeguard brain tissue beyond its known effect of reducing acute inflammation.
Check out this work published by the Pröbstel lab; Tradite Neziraj, Elisabeth Pößnecker and Anne-Katrin Pröbstel.
Congratulations to all the contributors for this excellent work!
Read full article here: https://www.science.org/doi/10.1126/scitranslmed.adi0295
Announcing: 1st-in-class anti-CD40L in multiple sclerosis
A significant advancement in the field of multiple sclerosis (MS) treatment! The research led by Patrick Vermersch, Cristina Granziera and Gavin Giovannoni surrounding the CD40–CD40L costimulatory pathway has paved the way for a potential game-changer - Frexalimab, a second-generation anti-CD40L monoclonal antibody.
The CD40–CD40L pathway plays a crucial role in regulating both adaptive and innate immune responses and has long been implicated in the pathogenesis of multiple sclerosis. With its intricate involvement in the disease process, targeting this pathway has become a promising avenue for developing effective treatments. Frexalimab represents a new frontier in MS therapeutics. As a second-generation anti-CD40L monoclonal antibody, it holds the potential to provide novel and improved solutions for individuals affected by this complex neurological disorder.
Multiple sclerosis manifests in two principal forms: relapsing and progressive. Relapsing MS is characterized by recurrent attacks, initially with remissions, but can lead to lasting disability over time. On the other hand, progressive MS involves continuous worsening. Understanding these distinct forms has been essential in tailoring effective treatment approaches. Relapsing multiple sclerosis primarily involves the adaptive immune system, orchestrated by circulating B lymphocytes that traffic into the central nervous system. In contrast, progression is mediated by both the adaptive and innate immune systems. The editorial accompanying this research by Stephen L. Hauser emphasizes the significance of addressing these different mechanisms in the treatment of two forms of MS. https://www.nejm.org/doi/full/10.1056/NEJMe2314434
The future of MS treatment is on the horizon, and we are eager to witness the positive changes that may result from this innovative research.
Read the full original article here: https://www.nejm.org/doi/pdf/10.1056/NEJMoa2309439?articleTools=true
Complement Activation Is Associated With Disease Severity in Multiple Sclerosis
Exciting news about the latest research article, "Complement activation is associated with disease severity in multiple sclerosis," published in Neurology: Neuroimmunology & Neuroinflammation authored by our esteemed team of researchers Johanna Oechtering, Sabine Schädelin, Jan Lünnemann and Jens Kuhle.
This study shows that the activation of the complement system, a component of the innate immune system, is significantly heightened in the cerebrospinal fluid (CSF) of individuals with a clinically isolated syndrome (CIS) and MS. Additionally, it has demonstrated a strong association between increased complement activation and the presence of an intrathecal IgM production.
It also uncovers a compelling correlation between elevated complement activation levels within the CSF and key clinical parameters including Expanded Disability Status Scale (EDSS) scores, future Multiple Sclerosis Severity Scores (MSSS), and Neurofilament Light (NfL) levels. These associations support the notion that complement activation may play a pivotal role in the pathology and progression of MS.
What does this mean for the future of MS treatment? This research suggests that complement inhibition could represent a novel and promising therapeutic target to mitigate disease activity and severity. By targeting complement activation, we may unlock new avenues for more effective MS management and ultimately improve the quality of life for individuals living with this challenging condition.
Congratulations to the dedicated @RC2NB team of researchers for their unwavering commitment to advancing science and making a meaningful difference in the lives of MS patients.
Read full article here.
Clinical Research Day Highlights: Advancements in Multiple Sclerosis (MS) Research

Last week, the Department of Clinical Research organized a Clinical Research Day to showcase the latest achievements in clinical research. The event aimed to highlight the work of the department's talented researchers, including some of our very own RC2NB team comprising Perrine Janiaud, Jens Kuhle, Lars Hemkens and Özgür Yaldizli. One of the standout talks during the event was presented by Perrine Janiaud, shedding light on significant progress in Multiple Sclerosis (MS) research.
Talk Summary: Leveraging sNfL for Personalized MS Care
Perrine discussed using serum neurofilament light chain (sNfL) as a sensitive marker for neuronal damage in MS. This marker provides valuable insights into disease activity and treatment response, offering a promising avenue for tailoring treatment decisions to individual patients with MS.
Through a Delphi study involving international MS experts and patient consultants, a consensus was achieved on treatment escalation, switching, and de-escalation algorithms based on sNfL levels. This consensus marks a significant step towards more personalized care for individuals with MS.
The next phase involves implementing these algorithms in a randomized pragmatic clinical trial known as the MultiSCRIPT trial. This trial is embedded within the Swiss MS cohort and assesses the benefits of 6-monthly sNfL monitoring compared to usual care. The trial aims to provide further evidence and insights into the potential advantages of incorporating sNfL monitoring into routine clinical practices.
Stay tuned for more updates on the MultiSCRIPT trial as the RC2NB team continues its efforts to advance personalized care for individuals with MS. The strides made in this research not only contribute to the scientific understanding of MS but also hold the promise of improving the lives of those affected by this complex neurological condition.
Image: Perrine Janiaud presenting during the DKF Clinical Research Day
Unlocking Real-World Insights in Multiple Sclerosis: Findings in Pragmatic Trials
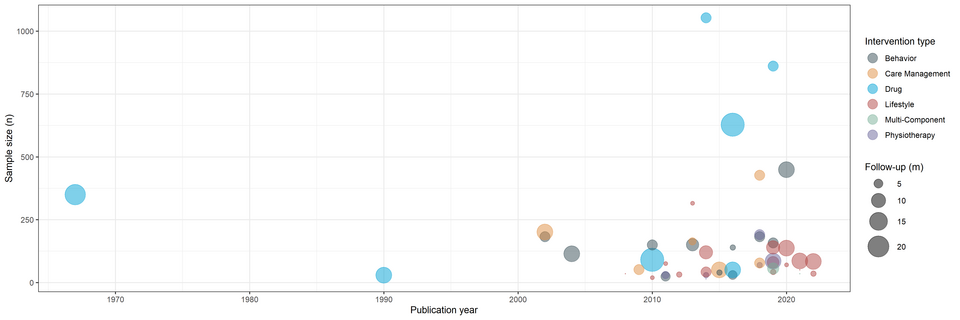
RC2NB is starting 2024 strong with the publication of a new scientific article that sheds light on the world of pragmatic trials and their crucial role in providing real-world evidence for treatment decisions in Multiple Sclerosis (MS).
The study led by Julian Hirt, Perrine Janiaud, Özgür Yaldizli, Ludwig Kappos, Cristina Granziera and Lars Hemkens aimed to discover the landscape of pragmatic clinical trials in multiple sclerosis (MS) with this systematic overview led by the Pragmatic Evidence Lab (https://pragmatic-evidence.org/) at RC2NB. A comprehensive analysis was conducted on 48 pragmatic trials from 1967 to 2022, examining their characteristics, demographics, and interventions.
The article emphasizes the urgency for the scientific community to come together and leverage the untapped potential of pragmatic trials in providing meaningful, real-world evidence for the advancement of MS treatment strategies. This research is a call to action to conduct more pragmatic trials and utilize routine data infrastructures for better results.
Do you want to know more? Check out the publication here
Happy Holidays 2023!

Dear Friends, Colleagues, and Supporters,
As 2023 comes to a close, we would like to take a moment to wish you and your loved ones a very happy Holiday Season.
Our objective is to advance neuroscience and enhance the quality of life for people suffering from Multiple Sclerosis and other neuroimmune diseases. We are proud to say that we have made significant progress in this direction, and we could not have done it without your invaluable support. We are deeply grateful to you all, and we thank you from our hearts.
Once again, we wish you Happy Holidays, and we are eagerly looking forward to the upcoming year 2024!
Warmest regards,
Your RC2NB Team
New Study Unveils Breakthroughs in Multiple Sclerosis Diagnosis: Diagnostic Performance of Cortical Lesions and Central Vein Sign
A recent study titled "Diagnostic Performance of Cortical Lesions and the Central Vein Sign in Multiple Sclerosis" has been published in the prestigious Journal of the American Medical Association (JAMA) Neurology. The study, led by researchers Dr. Alessandro Cagol, Prof. Cristina Granziera, and Prof. Ludwig Kappos contributes to the field of neurology by providing clinicians with better tools and insights for more accurate differentiation of multiple sclerosis (MS) from other neurological conditions that exhibit brain lesions on magnetic resonance imaging (MRI).
The study analyzes data from 1051 participants from 14 European centers within the MAGNIMS consortium. The participants included those with a diagnosis of MS, clinically isolated syndrome (CIS), or non-MS conditions associated with white matter lesions.
This research has set a major milestone in the diagnosis of MS as it provides strong evidence for the diagnostic use of these specific biomarkers for MSTo explore the diagnostic performance of Cortical Lesions (CLs) and the Central Vein Sign (CVS), in isolation and in combination was investigated for various cutoffs. A random forest model was used to rank the diagnostic importance of CLs and CVS compared to conventional MRI features, such as the presence of infratentorial, periventricular, and juxtacortical white matter lesions (WMLs). The results indicated that both CVS and CLs exhibited superior performance in comparison to conventional MRI features, aiding in the diagnosis of MS. The diagnostic performance of the CVS exceeded that of CLs and was increased when combining these two markers.
For more details, please refer to the full publication here.
SNF Sinergia Grant for Professors Tobias Derfuss, Christian Münz, and Burkhard Becher

Professors Tobias Derfuss, Christian Münz, and Burkhard Becher have received the Swiss National Foundation (SNF) Sinergia Grant for their research proposal on the role of B cells and EBV infection on Multiple Sclerosis. The interdisciplinary team of researchers from the University of Basel/University Hospital Basel and the Institute of Experimental Immunology Zurich, aim to investigate the mechanism by which Epstein Barr Virus (EBV) infection on B cells contributes to the formation of inflammatory lesions in the central nervous system (CNS) that cause symptoms of Multiple Sclerosis (MS).
The interdisciplinary team will use a systematic approach to study infected and non-infected B cells from MS patients and conduct hypothesis-driven experiments using a preclinical model with reconstituted human immune systems (humanized mice). The experiments seek to answer questions about the effects of EBV infection on the phenotype and function of circulating B cells, the impact on the response of autoreactive B cells when they encounter self-antigens, and if such virus-specific, infected or autoreactive B cells present myelin antigens to T cells to promote CNS autoimmunity.
This research, made possible by the Sinergia grant, will promote interdisciplinary collaboration and could lead to a better understanding of the causes of MS, potentially leading to new treatment options.
Two professors from University Hospital Basel, Ludwig Kappos and Jens Kuhle, on Clarivate's 2023 list of Highly Cited Researchers.

This prestigious recognition is awarded to influential researchers worldwide who have demonstrated significant impact in their fields of research. The selection process for the Highly Cited Researchers list is rigorous, relying on data from the Web of Science citation index and analysis conducted by bibliometric experts and data scientists at the ISI at Clarivate.
Prof. Tobias Derfuss Named an Honorary Member of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS).

We are extremely proud to share that Professor Tobias Derfuss has been named an Honorary Member of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS). This is a prestigious recognition that was officially announced during the ECTRIMS Congress in October 2023.
Prof. Derfuss's outstanding contributions to the field of Multiple Sclerosis and his dedicated service to the ECTRIMS over the years have earned him this well-deserved recognition. During the award ceremony at ECTRIMS 2023, Dr. Mar Tintoré, the ECTRIMS President, presented Prof. Derfuss with the award.
Prof. Derfuss has been a member of the ECTRIMS Executive Committee for eight years, from 2014 to 2022, serving as a treasurer, secretary general, and member. His dedication and leadership have made a significant impact on the ECTRIMS and MS community.
We are thrilled to celebrate this remarkable achievement with Prof. Derfuss and extend our heartfelt congratulations to him!
Image: Prof. Tobias Derfuss (right) and Dr. Mar Tintoré (left), ECTRIMS president, who presented him with the award.
Ocrelizumab exposure in relapsing–remitting multiple sclerosis: 10-year analysis of the phase 2 randomized clinical trial and its extension
We are excited to share the findings of a comprehensive study that sheds light on the safety and effectiveness of ocrelizumab in treating relapsing-remitting multiple sclerosis (RRMS) over an extended period. This is the longest follow-up study of ocrelizumab-treated patients with RRMS, spanning from 2008 to 2020.
The study confirmed the sustained efficacy of long-term ocrelizumab, with no new safety concerns identified. It's remarkable to note that even after longer interruptions of 6-month dosing cycles, most participants still maintained a reduced level of disease activity, and there was no evidence of rebound.
These findings offer hope to individuals living with RRMS, bringing us one step closer to enhancing their quality of life. With ocrelizumab's proven safety and effectiveness over an extensive follow-up period, we remain confident about its positive impact on the future of MS treatment.
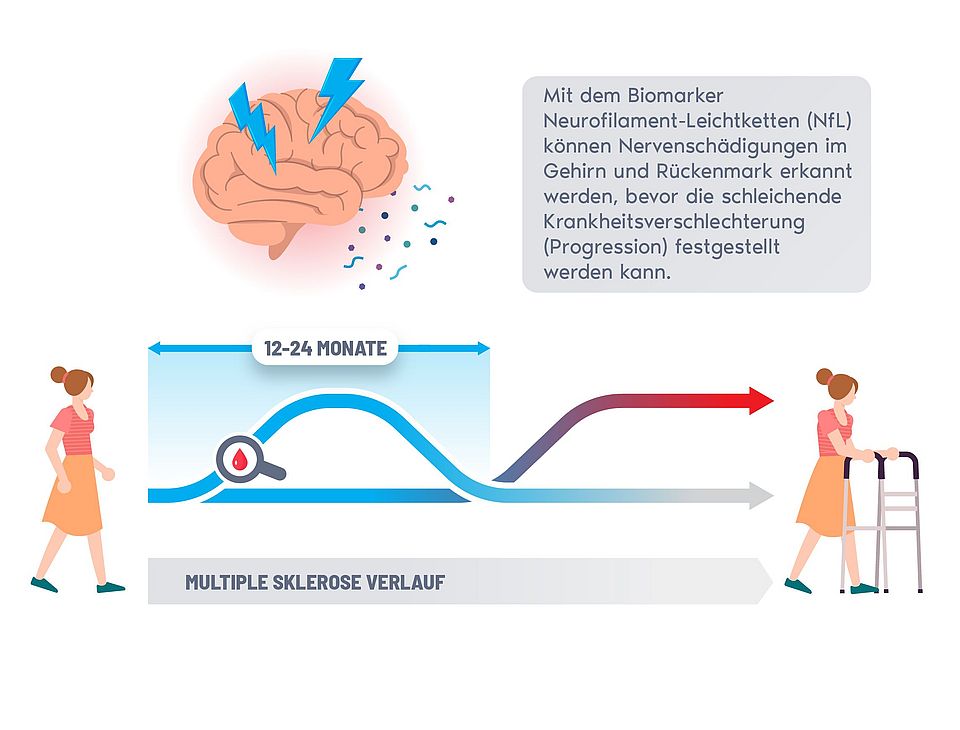
Press Release: Blood marker recognizes nerve injury in MS patients two years before clinical diagnosis and opens new treatment windows
Research project of the University of Basel and the University of California San Francisco published in “JAMA Neurology”.
Basel / San Francisco, 6th November 2023
Neuroscientists of the EPIC-Team (Expression, Proteomics, Imaging, Clinical) at the University of California San Francisco (UCSF) led by Ahmed Abdelhak, MD, and of the Swiss Multiple Sclerosis Cohort (SMSC) of the University of Basel under the leadership of Professor Jens Kuhle have conducted a study in multiple sclerosis (MS) patients that demonstrates that injury to the central nervous system can be detected up to 26 months before deterioration of neurological functions manifests clinically. The results of this study will be published in the renowned journal "JAMA Neurology" on November 6, 2023.
Neurofilament Light Chain (NfL) levels in the blood is a specific biomarker for damage of nerve cells and has been established earlier for monitoring acute disease activity and the efficacy of therapy. However, there was no biofluid marker defined that associates with continuous worsening of neurological functions, called 'progression', that occurs despite optimal therapy in most MS patients. The key finding of this study is that increased levels of NfL prognosticate progression in a period when it cannot be diagnosed with current clinical tools. This advance in early detection of disease course opens new treatment windows and better management of MS.
The study conducted by UCSF and University of Basel researchers aimed at addressing the current diagnostic gap in predicting disease progression in MS by analyzing long-term data from nearly 13,000 patient visits and blood samples. "The result that blood levels of NfL can prognosticate disease progression up to more than two years in advance of the occurrence of clinical manifestation of symptoms is a game changer for how we can monitor and manage the treatment of MS patients: it provides crucial rational for early therapeutic interventions," said Ahmed Abdelhak, the first author of the study.
The study demonstrates that NfL concentrations increase sharply between 12 and 26 months before the diagnosis of confirmed disability progression is made. Once the disability becomes clinically evident, NfL concentrations are no longer elevated compared to patients with stable disease, suggesting that this biomarker shows dynamic changes indicative of actual disease processes. This suggests that NfL measurements indicate underlying nerve injury earlier than physical examinations of neurologists. Overall, these findings can help doctors and clinicians identify patients who are at high risk of progression and provide early therapeutic intervention to contain disease progression.
Relevance for MS-Research
This study combines two of the largest and best documented MS cohorts worldwide. It builds on earlier work conducted by researchers from the University of Basel that were published in February 2022 and September 2023 in “Lancet Neurology”. The Swiss researchers demonstrated therein the added value of NfL as a blood biomarker for assessing the disease activity state of MS patients by establishing normative data for this biomarker in adults and children that allows to qualify NfL concentrations as normal or increased.
„Besides the groundbreaking insights regarding the temporal link between NfL elevation and disease progression in MS, the study reinforces the pivotal role of NfL as an early marker of neuronal injury. Monitoring NfL levels has already been established as an important indicator to detect concurrent disease activity with higher sensitivity than a clinical examination or conventional MRI scans. The current study further stresses the critical value of NfL measurement for a more personalized medical treatment”, says Professor Jens Kuhle.
The focus of future studies is how the new insights on what increased NfL means can be translated into better treatment algorithms.
Relevance for MS Patients
Nearly three million people worldwide suffer from MS. Every progress in the understanding of this disease has the potential to improve therapy efficacy, and hence the lives of patients as well as their families. Early and precise detection of prospective damages opens new windows of early therapeutic intervention. The therapeutic target is to hold NfL concentrations at levels of healthy persons.
CLINNOVA: A Trans-Regional Digital Health Effort Unlocking the Potential of Artificial Intelligence and Data Science in Health Care – Use Case Multiple Sclerosis

Background
Multiple sclerosis (MS) is the most common immunological disorder of the nervous system (> 1 in 1000). This disease has a strong female preponderance and is the most common cause of permanent disability in young adults. MS affects the central nervous system causing a variety of neurological symptoms escalating disability over time. Despite the availability of a large panel of approved disease-modifying MS drugs, frequently physicians lack data-based support to prescribe the most effective treatment at a given time point along the patient pathway, highlighting the need for personalized clinical management. Artificial intelligence (AI)-driven solutions hold a high potential for precision medicine but have not yet fulfilled expectations. Thanks to a trans-regional collaboration between institutions from four countries (Switzerland, Germany, France, and, Luxembourg), CLINNOVA has the mission to support the digitalization of healthcare, unlock healthcare’s AI potential, and improve personalized treatments of chronic inflammatory diseases. The CLINNOVA consortium focuses on 3 Use Cases of immune-related diseases with large unmet medical burden: multiple sclerosis, inflammatory bowel diseases and, rheumatoid diseases.
Research Question
CLINNOVA-MS aims to build a prospective MS patient cohort across the participating clinical centers to generate structured, standardized, interoperable, quality-controlled, multi-sourced, and multi-dimensional data. These data will be analyzed in a federated learning platform to train and develop AI-algorithms generating personalized therapy solutions for patients, identify characteristics associated with early MS and transitioning phases to progressive MS, and validate digital biomarkers allowing a continuous monitoring of those patients.
Study Methodology
CLINNOVA-MS is a prospective cohort study including participants with early MS or in the transitioning phase to progressive MS. Up to 100 participants will be enrolled in the study in Basel and an equivalent number should be also enrolled at each participating center.Eligible patients will be asked to provide data and samples and will be followed up for 5 years starting from the date of inclusion. Patients reported outcomes and cognitive and motor assessments will be performed using the Healios+Me smartphone application. Biological samples (i.e., blood, cerebrospinal fluid, and stool) as well as imaging data (if performed as per standard of care) will be collected locally, then processed and analyzed centrally at the appropriate partner institutions. The sample-associated data, monitoring data collected via the digital application and patient visits data will be analyzed through a privacy-preserving platform using federated learning. This approach allows to train algorithms on datasets from different clinical sites without sharing sensitive data.
Significance of the study
Thanks to its consortium, CLINNOVA-MS will make a significant step forward to facilitate early MS detection, enable personalized medicine approaches through AI‐based stratification, and improve patients’ care and quality of life while reducing healthcare costs.
Contributors
Prof. Ludwig Kappos (RC2NB, USB)
PD Dr. Lars G. Hemkens, MPH (RC2NB, USB)
Dr. Bebeka Cosandey (RC2NB, USB)
Dr. Amandine Bovay (University of Basel)
Dr. Bram Stieltjes (University Hospital Basel)
Dr. Thierry Sengstag (University of Basel)
Read Full DKForum Article Here (German)
Revolutionizing MS Patient Care with Smartwatch Technology
We are thrilled to announce the release of the latest research paper that delves into the potential of smartwatches as an effective tool for monitoring the health of individuals with Multiple Sclerosis (MS). The paper, titled "Smartwatch-Derived Features for Assessing Health in Multiple Sclerosis Patients," has been published in the journal of Multiple Sclerosis and Related Disorders and presents significant findings that could change how we assess the well-being of MS patients.
The future of healthcare is here thanks to the research of Dr. Tim Woelfle, Silvan Pless, Dr. Johannes Lorscheider, Dr. Andrea Wiencierz, Prof. Cristina Granziera, and Prof. Ludwig Kappos.
Key findings from recent research include:
- Smartwatches can reliably measure activity, sleep, and heart rate features.
- Smartwatch-derived features can effectively distinguish between individuals with MS and healthy volunteers, especially those with moderate disability.
- There are strong correlations between smartwatch data and three clinical reference tests, including the Expanded Disability Status Scale (EDSS).
- Sleep and heart rate data add value to step counts in assessing MS patients' health.
This research brings us closer to a more personalized and effective approach to MS care. By leveraging wearable technology, we can provide patients with the support and insights they need to manage their condition more effectively.
Continuous progression independent of relapse activity (PIRA) occurs in all stages of MS. Uniform diagnostic criteria published
Multiple sclerosis is typically characterized by relapses, i.e. neurological dysfunctions occurring within days, followed by more or less complete recovery. It was assumed that in the time between the relapses, the disease is dormant and no deterioration occurs. Only later in the course, usually after 10-20 years, does the progressive phase follow in most affected persons, with a more continuous increase in disability. However, systematic long-term studies and the use of increasingly effective drugs to prevent relapses have shown in recent years that continuous progression also occurs in patients with a typical relapsing course. This progression independent of relapse activity (PIRA) is responsible for the majority of the increase in disability from the earliest stages of MS. After the first descriptions of PIRA, numerous studies in different patient groups followed with the aim of better understanding this newly recognized phenomenon.
Until now, however, there was no uniform definition of PIRA, which made it difficult to compare and interpret the studies. Researchers from the Research Center for Clinical Neuroimmunology and Neuroscience (RC2NB) led by Jannis Müller, Cristina Granziera and Ludwig Kappos, who were already significantly involved in the initial description of PIRA, have now summarised the current state of knowledge about PIRA in a systematic review in the renowned journal JAMA Neurology and proposed uniform diagnostic criteria based on this review. Such uniform diagnostic criteria create better conditions for the early and reliable detection of PIRA and for the prevention of PIRA by means of targeted, individually tailored therapies.
View Abstract on JAMA Neurology.
16 October 2023
Possible biomarker of MS-like autoimmune disease discovered (Pröbstel Lab)
It has been known for several years that the diagnosis “multiple sclerosis” conceals a whole range of different illnesses, each requiring customized treatment. Researchers at the University of Basel and the University Hospital of Basel have now described a possible new MS-like disease and explained how to diagnose it.
Multiple sclerosis (MS) is characterized by areas of inflammation in the central nervous system. The immune system attacks the body’s own structures and destroys the covering of nerve cells, known as the myelin sheath. The picture research has painted of the illness is somewhat more complicated, however. It can cause a variety of neurological symptoms, like paresthesia and paralysis of the limbs, which progressively or abruptly worsen. Which parts of the nervous system are affected varies substantially between individuals. A treatment may work for some patients – but make the condition worse in others.
“There’s a huge amount of diversity in how inflammatory autoimmune diseases of the central nervous system like multiple sclerosis present,” explains Professor Anne-Katrin Pröbstel of the University of Basel and University Hospital Basel. Researchers have been gradually discovering the key distinctive features of “atypical” cases of MS for the past ten years. A few of these autoimmune diseases have been given different names to better distinguish them from MS even though they also destroy the myelin sheath. Victims of these diseases often have inflammation in their spinal cords or optic nerves.
In a study of roughly 1,300 patients, Pröbstel’s team has now discovered a biomarker that may make it possible to differentiate another MS-like illness from the others. The researchers have reported their findings in the journal JAMA Neurology.
View Full Article.
Wearable Sensor Technologies to Assess Motor Functions in People With Multiple Sclerosis: Systematic Scoping Review and Perspective
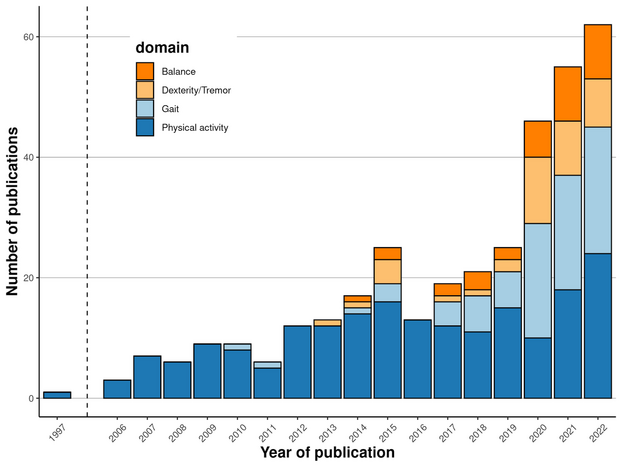
Our systematic scoping review and perspective "Wearable Sensor Technologies to Assess Motor Functions in People With Multiple Sclerosis" has just been published in the Journal of Medical Internet Research. We have included 308 papers from 1997 to 2022 with different contexts: real-world, laboratory, and mixed. They assessed physical activity, gait, dexterity / tremor, and balance. Sensors included accelerometers, gyroscopes, magnetometers, touchscreens, and mechanical pedometers. We believe quasi-continuous remote monitoring of motor functions could help optimize the disease management in the future.
The accompanying web app allows interactive result exploration and the local citation network shows the connections between the 308 included papers and their co-authors.
Woelfle T, Bourguignon L, Lorscheider J, Kappos L, Naegelin Y, Jutzeler CR. Wearable Sensor Technologies to Assess Motor Functions in People With Multiple Sclerosis: Systematic Scoping Review and Perspective. J Med Internet Res 2023;25:e44428
View Full Article on PubMed.
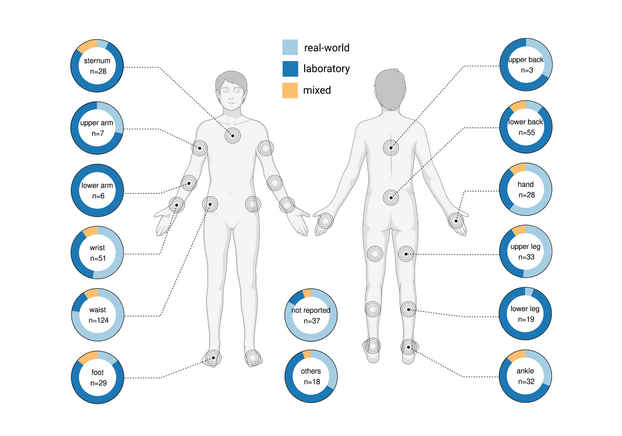
Study increases precision of assessment of nerve damage in children
International research collaboration defines age-specific reference range for blood neurofilament light chain in pediatric health.
Blood Neurofilament Light Chain (NfL) is an innovative biomarker that specifically indicates neuroaxonal injury. However, its clinical application has been constrained due to the lack of established reference ranges for children and adolescents. Accurate monitoring of neuroaxonal injury in neurologic and systemic diseases is of pivotal importance at population and individual patient levels. In the paediatric population, tools capturing neuroaxonal injury with high specificity would substantially facilitate early and accurate detection of conditions associated with short- and long-term neurological disabilities with considerable socioeconomic impact. This may as well accelerate clinical trials evaluating the expanding library of targeted causal and disease-modifying treatments.
The Study
Led by Jens Kuhle at the University Hospital of Basel and Sven Wellmann at the University Hospital of Regensburg and an international team comprising top-tier institutes from Switzerland, Germany, and the United States conducted comprehensive analyses of NfL concentrations in more than 2,500 healthy children and adolescents. This ground-breaking study has been published in the esteemed journal, The Lancet Neurology.
View Full Article on Lancet Neurology.
28 July 2023
Interview with Prof. Kuhle on the Swiss Multiple Sclerosis Cohort (SMSC) and blood biomarkers
Published on 17 July 2023 on Medical Tribune.
Article available in German only.
Is sGFAP a Dynamic New Biomarker for Disease Progression in MS?
— An international team of experts certainly thinks so. Their new findings indicate that serum glial fibrillary acidic protein can predict worsening independent of relapse activity in multiple sclerosis.
A serum biomarker for disease progression in multiple sclerosis (MS) may be a game changer for research and, ultimately, for treatment of patients with the most devastating forms of the disease, indicate the results of a new study.
View Full Article on MEDPAGE TODAY published on 11 May 2023.
Ellermann Prize 2022
Congratulations to Dr Pascal Benkert, Head of SMSC datacenter, and Stephanie Meier, candPhD, both members of Prof Jens Kuhle’s research group at Neurology/RC2NB who have been honored with the Ellermann Prize 2022 by the Mogens und Wilhelm Ellermann-Stiftung.
Their work published in Lancet Neurology demonstrated how concentrations of serum neurofilament light chain (sNfL) are a sensitive measure of disease activity in multiple sclerosis (MS). By creating a large reference database from control persons they were able to show that patients with increased levels of this structural neuronal protein in blood experience more disease activity in the next year based on two large cohorts of patients followed in the Swedish MS Registry and the Swiss MS Cohort Study. sNfL has meanwhile increasingly entered clinical care of MS patients.
Annual Report 2022
We are proud to publish our Annual Report 2022.
2022 has been another year of transformation and growth for RC2NB. Important milestones were achieved across all RC2NB’s research groups and a new 4th workstream “Pragmatic Trials and Real World Evidence” now complements the other three.
Please find the link to the Report here. If you would like to receive a hardcopy, please contact us.
Swiss Multiple Sclerosis Society Award
Congratulations to Prof. Jens Kuhle and Prof. Tobias Derfuss, two RC2NB researches, who have been honored with an award by the Swiss Multiple Sclerosis Society. The recognition, endowed with 100,000 Swiss francs, goes to both of them for their exceptional contributions to MS research and their great commitment to those affected by the disease.
Both work in neurology at the University Hospital Basel (USB) and collaborate closely with each other at the RC2NB. They both sit on the medical-scientific advisory board of the Swiss MS Society.
Read more on the Medinside Website.
Last updated 31 Jan 2023
Bing Research Prize 2022
Congratulations to Prof. Cristina Granziera, Workstream Leader and Deputy Head of RC2NB for receiving the highly prestigious Bing Research Prize 2022 awarded by the Swiss Academy of Medical Sciences.
The Prize was awarded in appreciation of her important contributions in the interface of clinical neurology, neuroradiology and medical physics. Applying highly innovative, advanced neuroimaging methods, allows her to better characterize the changes occurring in the brain and spinal cord of patients suffering from multiple sclerosis and to identify biomarkers that offer a very promising foundation for novel diagnostic procedures. Prof. Granziera’s results, internationally recognized and validated in large longitudinal clinical studies, have a direct impact on the treatment and follow-up of patients with multiple sclerosis.
Last updated 31 Jan 2023
Clarivate Highly Cited Researchers™ list 2022
With Ludwig Kappos and Jens Kuhle two RC2NB researchers have been named on the Clarivate Highly Cited Researchers™ list 2022.
The list identifies researchers based on the citation counts of their work in the last ten years, indicating the impact of their work. Clarivate™ states that “Of the world’s population of scientists and social scientists, Highly Cited Researchers™ are 1 in 1,000.”
In 2022, Clarivate™ counts a total of 6938 researchers from 21 research fields and 69 countries.
A blood marker for multiple sclerosis progression
For the third time in a row, Jens Kuhle, Head of Multiple Sclerosis Centre at the University Hospital Basel and DKF/DBM research group leader, is awarded an SNF project grant for the development of biomarkers for the detection and prognosis of disease activity in multiple sclerosis (MS) on a personalised level.
Read more on the Department of Clinical Research website.
DreaMS study opened for clinical study recruitment and reached first milestone
More than 50 people with multiple sclerosis support the development of the novel dreaMS digital health app, by participating in a clinical validation study led by the Research Center for Clinical Neuroimmunology and Neuroscience Basel (RC2NB).
The dreaMS app provides so-called digital biomarkers, measurements of health status and disease progression collected in the usual everyday life in a real-world environment by using a smartphone. Digital biomarkers aim to detect subtle disease progression earlier and more comprehensively, thus complementing standard assessments by neurologists, laboratory or neuroradiological examinations. The dreaMS app is part of a digital platform for optimized disease management to better involve patients and improve the communication with their health care team. DreaMS and the digital platform are developed jointly by the RC2NB and Healios AG, Basel.
The DreaMS validation study started in April 2022, and with the enrollment of 50 patients, has now reached an important milestone.
Participants in the dreaMS study install the dreaMS app on their own smartphone and digital biomarkers are then collected over a period of 2 years. This digital information will be compared with state of the art established clinical, imaging, and body fluid assessments obtained in the framework of the Swiss MS Cohort Study over the study period. Good agreement would indicate high validity of the digital biomarkers. Digital biomarkers are expected to allow earlier and more comprehensible detection of subtle disease progression and in the future allow to tailor treatment more individually.
PD Dr Johannes Lorscheider, PI of this study, said, ‘The DreaMS validation study will provide valuable information on the reliability and accuracy of monitoring disease evolution and treatment response with a digital app. With validated digital biomarkers, we hope to further improve the care of our patients.’
Professor Ludwig Kappos, director of the RC2NB said, ‘We are very grateful to all our collaborators, funders and partners who are supporting us at RC2NB to explore this fascinating future field and to find and establish generally accepted digital measures for better care of persons with MS and other neurological diseases’.
The news has also been published by the University Hospital Basel on their website.
Last updated 31 Oct 2022
Last updated 31 Oct 2022
Reliability and acceptance of dreaMS, a software application for people with multiple sclerosis: a feasibility study
We just published the results of the first reliability and acceptance assessment of the dreaMS software application in the Journal of Neurology.
Woelfle T, Pless S, Reyes O, Wiencierz A, Feinstein A, Calabrese P, Gugleta K, Kappos L, Lorscheider J, Naegelin Y. Reliability and acceptance of dreaMS, a software application for people with multiple sclerosis: a feasibility study. J Neurol. 2022 Aug 30:1–10
[PubMed]
Last updated: 5 Sept 2022
High-dimensional immune profiling identifies a biomarker to monitor dimethyl fumarate response in multiple sclerosis
The paper "High-dimensional immune profiling identifies a biomarker to monitor dimethyl fumarate response in multiple sclerosis" has published in PNAS.
Understanding the immunobiology of multiple sclerosis remains an unresolved challenge. Distinctive features of therapeutic response to effective treatments are one important source of insight into the underlying pathomechanisms. Here, we studied dimethyl fumarate, a routinely administered but mechanistically poorly understood compound. We developed an approach using multipanel immunophenotyping by mass cytometry in conjunction with a weakly supervised machine-learning algorithm to facilitate unprejudiced identification of rare but specific immune cell populations accounting for beneficial treatment response. This approach allowed identification of a subset of antigen-experienced T helper cells that are preferentially depleted in responding individuals. As a biomarker, this subset may guide clinical stratification.
Last updated 9 Aug 2022
Last updated 16 May 2022
Last updated 17 Feb 2022